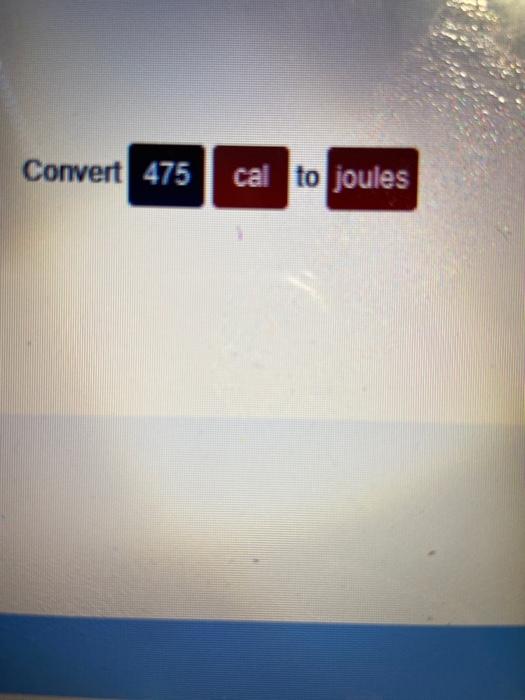
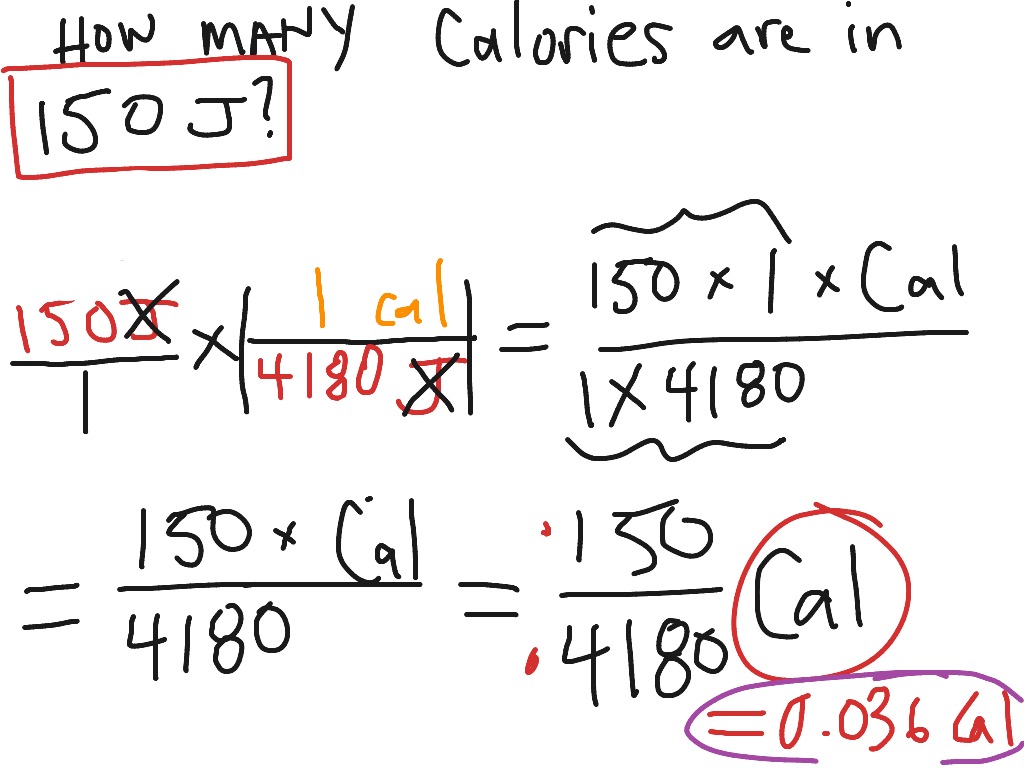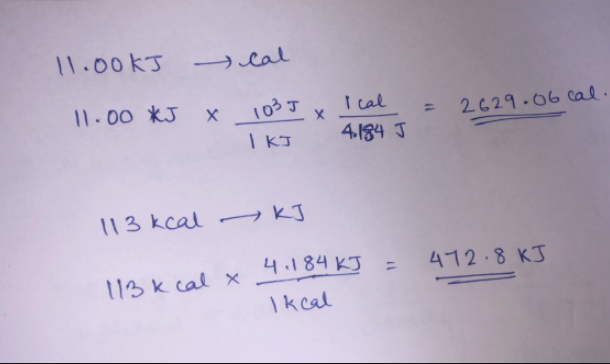SOLVED:Perform the indicated conversions. a. 85.21 cal into joules b. 672.1 \mathrm{J} into calories c. 8.921 \mathrm{kJ} into joules d. 556.3 cal into kilojoules
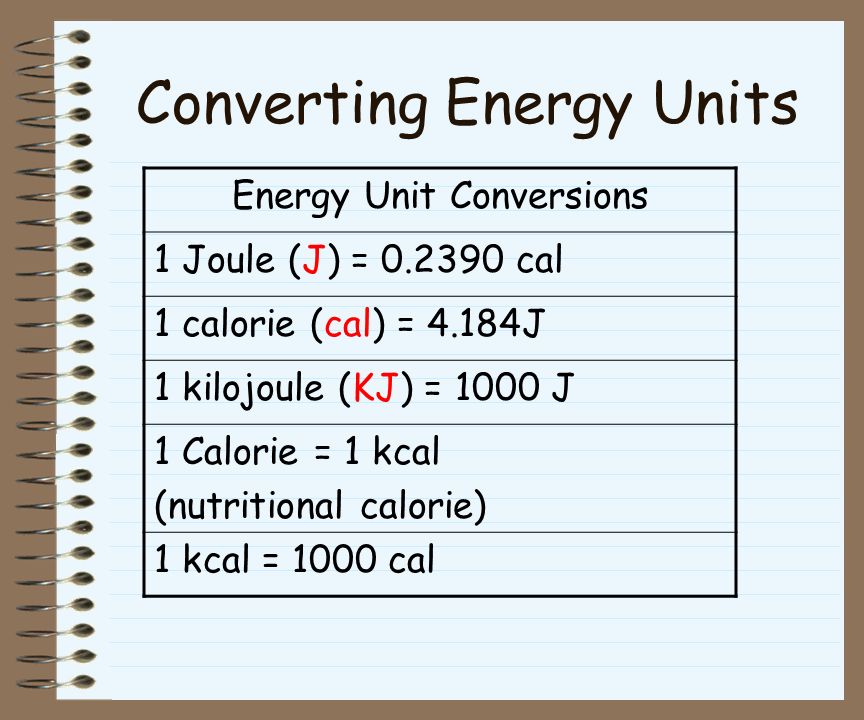
Energy Ability to “do work” or produce a change. Forms: potential energy and kinetic energy Types: mechanical, electrical, nuclear, solar, chemical, etc. - ppt download

6. Morning breakfast gives 5000 cal to a 60 kg person The efficiency of person is 30%. The height upto which the person can climb up by using energy obtained from breakfast

ENERGY & MATTER Chapter 2. Wednesday, 10/1/14 Learning Target: Know the 3 basic forms of energy and how energy is calculated. Learning Outcome: I will. - ppt download

The calorie is defined as `1 cal= 4.86 `joule Why as `1 cal= 4J` to make the conversions easy? - YouTube

SOLVED:Perform each conversion. (a) 588 cal to joules (b) 17.4 J to Calories (c) 134 \mathrm{~kJ} to Calories (d) 56.2 Cal to joules

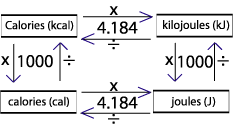
Energy (heat) may be expressed in joules or calories. 1 calorie (cal) = joules (J) How many joules in 60.1 calories? How many calories. - ppt download
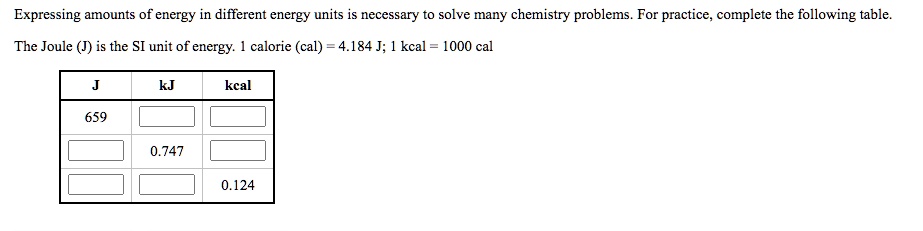
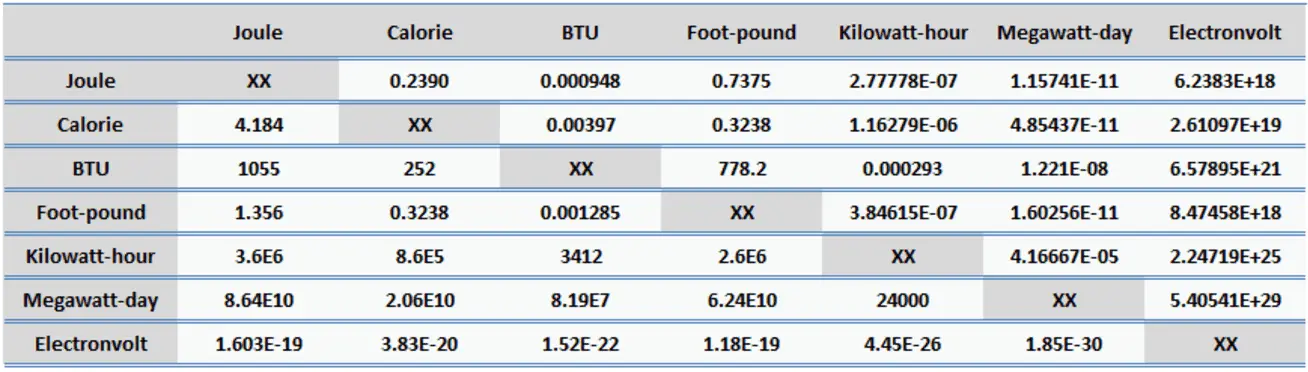
SOLVED:Expressing amounts of energy in different energy units is necessary to solve many chemistry problems. For practice, complete the following table_ The Joule (J) is the SI unit of energy: calorie (cal)

69. Energy equivalent to one erg, one calorie andone joule is in the order :(a) 1 Cal>1 Joule>1 Erg(b) 1 Erg>1 Cal>1 Joule(c) 1 Joule>1 Cal>1 Erg(d) 1 Cal>1 Erg>1 Joule | Snapsolve